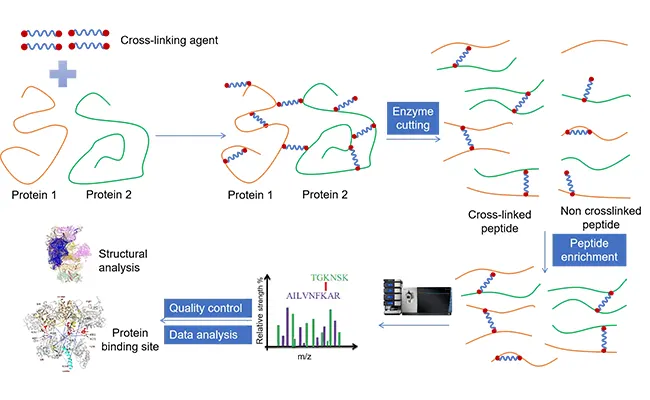
bài liên quan
RNA Extraction Sonicator: Cung cấp dữ liệu Proteomics có thể tái tạo mà không bị tổn thương do nhiệt
2026-04-13Proteomics researchers face a persistent sample preparation problem: the same heat that helps break cells also silently destroys the data they are trying to collect. A well-designed RNA Extraction Sonicator with true low-temperature focused ultrasound technology directly addresses this conflict.
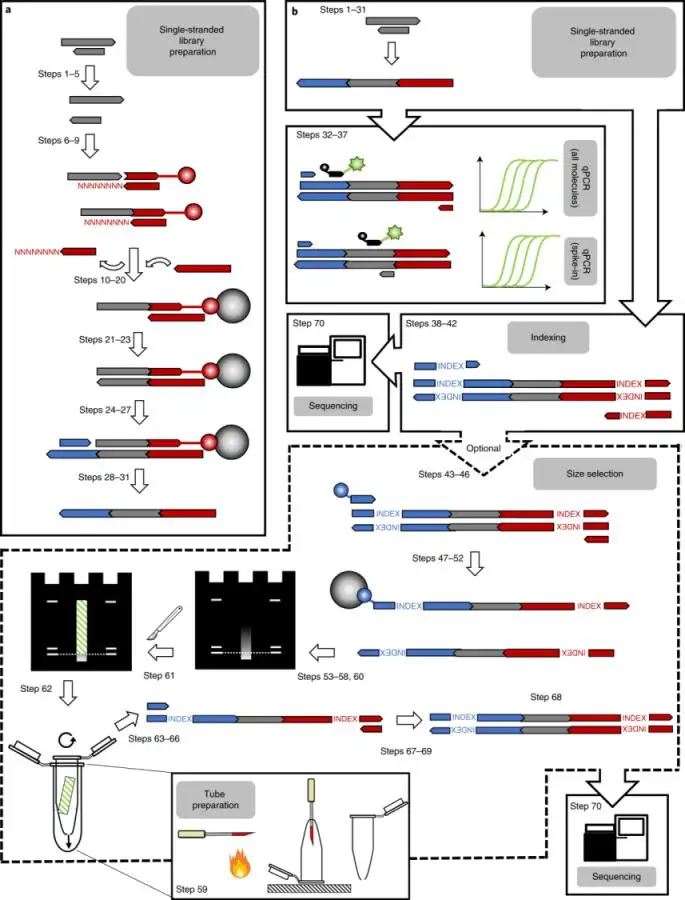
Manual and automated preparation of single-stranded DNA libraries for
the sequencing of DNA from ancient biological remains and other sources of highly degraded DNA
Uncontrolled heat during sonication does not just degrade biomolecules—it systematically erodes quantitative comparability across replicates, operators, and instruments. The way samples are disrupted marks the start of the journey to reproducible proteomics data. This journey involves neither superior mass spectrometers nor elaborate software.
Why Proteomics Reproducibility is Being Hampered by Heat
Many manufacturers of bath and probe sonicators did not design them keeping proteomics in mind. The mechanisms of sonicators and their caviatiton streams produce micro-medium thermal spikes. Generally, this leads to spikes in temperature, and as a result, denaturation of the protein. This denaturation accelerates the activities of proteases and nucleases, and introduces downstream biases in fragmentation. This rapid thermal instability makes sonicators favor the abundant, thermostable proteins and systematically reduce the detection of low-abundance or heat-sensitive targets. This disrupts the quantitative integrity of the results.
The problems we pose here are not purely hypothetical:
•Sample preparation is the bedrock: Lab bubble sonicators and other temperature-uncontrolled disruption equipment are not as useful when we can only control the lysis conditions of disruption to only introduce biases.
•The value of high-resolution mass spectrometers is, essentially, zero when reproducibility is introduced in workflows in the proteomics.
•When the conditions of fragmentation change, instrumentation drift signals and fading peaks, the results of biological discovery deteriorate, and troubleshooting becomes the focus, as opposed to answers.
A Focused Ultrasound Alternative
An RNA Extraction Sonicator utilizing the focused ultrasound design presents significant advantages over traditional probe or water bath systems for proteomic workflows. Unlike the water bath probe systems which inundate the sample with water and acoustic energy or which make direct probe contact with the sample, focused systems use high-frequency, short-wavelength ultrasound and contactless and isothermal water medium that focuses ultrasound energy at the sample vessel.
For proteomic workflows, this design has the following advantages:
•Reduces the variability caused by contact with the probe. The depth of insertion, alignment, and erosion of the probe tip will no longer affect how much energy is absorbed by the sample. This eliminates an important source of variability in inter-operator and inter-run settings.
•Low-temperature stability is preserved at the sample. An insulating medium focuses ultrasound in the sample area, and a delicate temperature feedback loop prevents and monitors the sample area from damage, as a result of overheating the sample and the nucleic acids, and prevents protein denaturation as a result of overheating.
A Focused Approach, Backed by Research
Much can be confirmed in the literature. An example of where focused ultrasonication was used was in Nature Protocols (2010), and as a further example, Meyer and Kircher described how it was possible to use focused ultrasonication to control thermal publisher bias and library complexity by providing an acoustic focal geometry and controlled temperature environment for multiplexed sequencing for short and tight DNA fragments. In the proteomics research focused on MALDI-TOF, they used focused ultrasonication to identify filamentous fungi at an operable time of less than 5 minutes per sample and maintained their protein extraction efficiency.
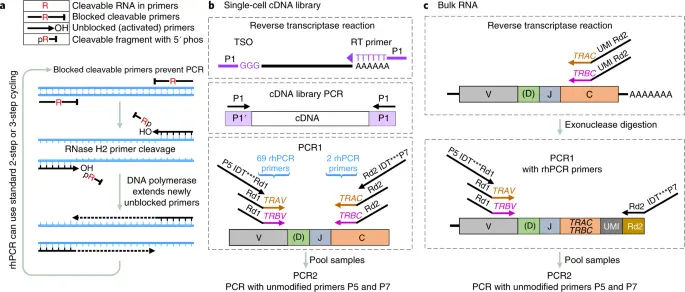
RNase H–dependent PCR-enabled T-cell receptor sequencing for
highly specific and efficient targeted sequencing of T-cell receptor mRNA for single-cell and repertoire analysis
On a larger scale, sample preparation for proteomics has been recognized as an important element in the quality and reproducibility of data in the domain. Whether working with clinical serum, fresh tissue, or difficult-to-lyse microorganisms, the principle holds: controlled, consistent disruption is a prerequisite for meaningful quantitative proteomics.
Building Reproducible Proteomics on a Foundation of Control
The translation from protein extraction to peptide identification depends on controlling variables—temperature, energy exposure, sample handling—at every stage. A RNA Extraction Sonicator that delivers non-contact, isothermal, focused acoustic energy removes two historically uncontrolled variables: thermal degradation and operator-dependent energy delivery.
When samples are processed under true low-temperature conditions:
•Protein conformation is preserved, minimizing artifactural degradation and ensuring that the extracted proteome accurately reflects the biological state of the sample.
•Enzymatic digestion steps proceed from a consistent starting point, reducing peptide-level variation that otherwise complicates quantification.
•Downstream mass spectrometry data exhibits lower coefficient of variation across replicates, supporting confident identification of both high-abundance and low-abundance targets.
Meeting the Demands of Modern Proteomics
Công nghệ Longlight's BoFU-80 Focused Ultrasonicator is engineered around a proprietary confocal ultrasonic transducer that concentrates acoustic energy precisely at the sample through an isothermal, non-contact water bath, safeguarding biomolecules from thermal stress throughout the sonication process. The system is fully calibrated before shipment to ensure uniform performance, and a highly sensitive temperature control system verifies that samples remain unaffected by incidental heat generation, greatly reducing errors introduced by manual operation.

The device aids diverse areas of proteomics research:
•Cell disruption and protein extraction to access both abundant and low-abundant microscale targets.
•Processing of FFPE samples for proteomic studies.
•Homogenization of firm and complex sample matrices.
•Preparation of samples for MALDI-TOF MS to identify microbes and clinical microbes.
Kết thúc
In the field of proteomics, reproducibility is related to the different choices of sample preparation, rather than an abstract, and consistency can be engineered with sample preparation. A RNA Extraction Sonicator with controlled and focused, temperature controlled, ultrasound, engineered consistency. Sample processing is no longer a burden, and consistency is replaced with variability. For environments—clinical diagnostics, pharmaceutical development, or fundamental research—where every replicate counts, that control is not merely convenient; it is essential.
Những câu hỏi thường gặp
Q: What measures are used to protect the sample from overheating during processing in an RNA Extraction Sonicator?
A: The sonicator uses focused ultrasonication to enforce an isotropic relationship between the sample and the damage that could occur from overheating.
Q: Is the same RNA Extraction Sonicator used in both proteomics and genomics workflows?
A: Yes, the sonicator can work in both proteomics and genomics workflows.
Q: What types of samples are optimized with the use of the low-temperature focused ultrasonicator?
A: low-temperature focused ultrasonicator best fit the sample type that are heat-sensitive in nature, low-abundance, or are difficult to conjure/lyse such as microorganisms and FFPE tissues.
Q: What is the shortfall of the focused ultrasonication in the clinicians lab?
A: The focused ultrasonication systems indeed have significant variability, especially when compared to traditional probe sonicators that greatly rely on user input.