bài liên quan
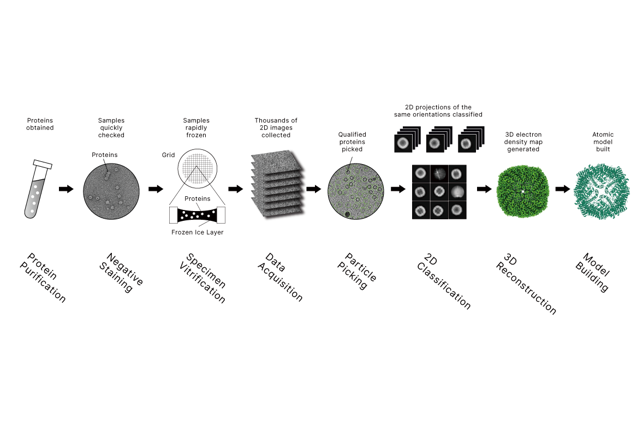
Cryo-EM độ phân giải cao cho các mục tiêu dưới 100 kDa: Cách tiếp cận mới, kết quả thực tế
2026-04-20High-Resolution Cryo-EM has become a routine method in structural biology, yet nearly 75% of human protein-coding genes produce proteins below 50 kDa—a segment that remains significantly underrepresented in the Electron Microscopy Data Bank (EMDB). This gap is not due to a lack of importance, but to a fundamental physical constraint: smaller particles generate inherently weaker signal relative to background noise, making them difficult to pick and align during image processing.
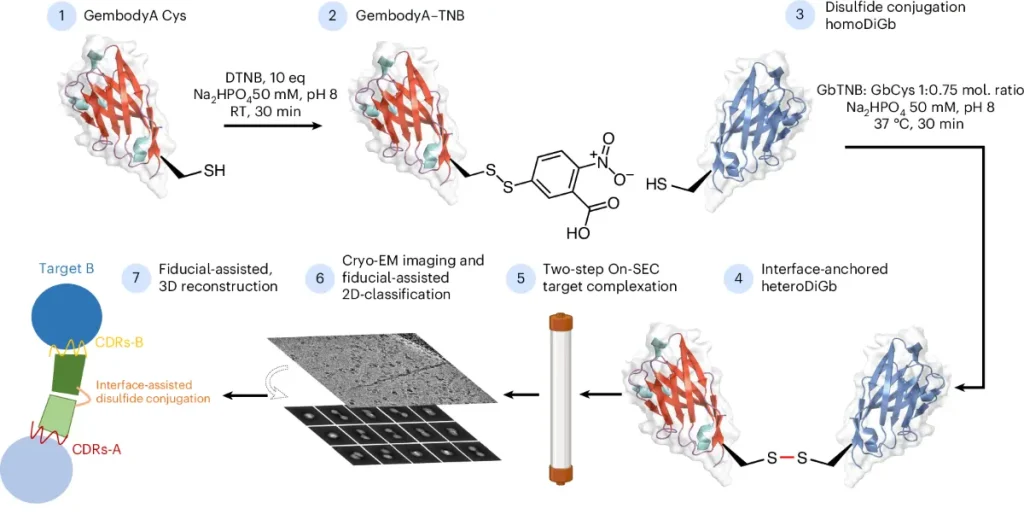
Covalently constrained 'Di-Gembodies' enable parallel structure solutions
by cryo-EM | Nature Chemical Biology
Over the past several years, we have been fortunate to witness a surge of practical applications and innovations, including engineered scaffolds, AI-optimized rigid tools, advanced tools, and more efficient and effective workflows. Here, we discuss the challenges of targeting sub-100 kDa, the newest approaches to address these challenges, and how these approaches have begun to be a part of standard practice.
Challenges of Small Proteins
Single particle cryo-EM works best for large size complexes. Signal is abundant and makes processes of alignment and reconstruction of three-dimensional models easier. However, for smaller targets, there are two persistent challenges:
• Lowers Signal to Noise Ratio: Smaller complexes scatter fewer of the electrons. Therefore, these targets become more challenging to separate the true signal from the background noise.
• Insufficient Structural Components: Small proteins do not have prominent features. As a result, algorithms for cryo-EM struggle with proper particle picking and proper orientation, causing high B-factors and reconstructions of poor quality.
These challenges account for the fact that less than 4% of the deposited structures that are a part of the EMDB are below 100 kDa, even though small proteins are abundant in both eukaryotic and prokaryotic organisms.
Techniques that Enable Cryo-EM to Address Small Protein Targets
Several separate approaches to solve the low-SNR bottleneck have been explored. Most of the approaches involve maintaining the native conformation of the small target proteins. The primary approach is to increase the effective size of the target proteins and complexes.
Mass-Enhancement Scaffolds: Plain and Simple
Binding proteins and protein-based scaffolds help add particle mass that improves alignment. Three outstanding scaffold designs that prove success include the following:
• Di-Gembodies (Nature Chemical Biology, 2025): Coupling constrained, covalent nanobody dimers, and engineered interface capture, this method enables virtually any scaffold protein structure determination, including the recent resolution of the 14 kDa hen egg white lysozyme cryo-EM structure, the smallest cryo-EM structure to date. Rosalind Franklin Institute, University of Oxford, and Diamond Light Source developed this modular approach, and there is no need for traditionally time-consuming re-optimization stepping for a new protein target.
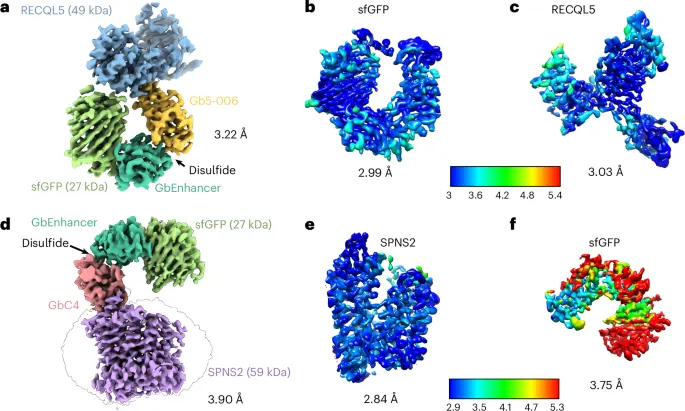
Covalently constrained 'Di-Gembodies' enable parallel structure solutions
by cryo-EM | Nature Chemical Biology
• DARPin-Apoferritin Scaffold (IUCrJ, 2025): Symmetrical, octahedral, and 1 megadalton apoferritin-designed scaffold proteins allowed for the crowding of the sample and cryo-EM near- and sub-atomic resolution (achieving a 70% increase modification in protein stiffness and alignment).
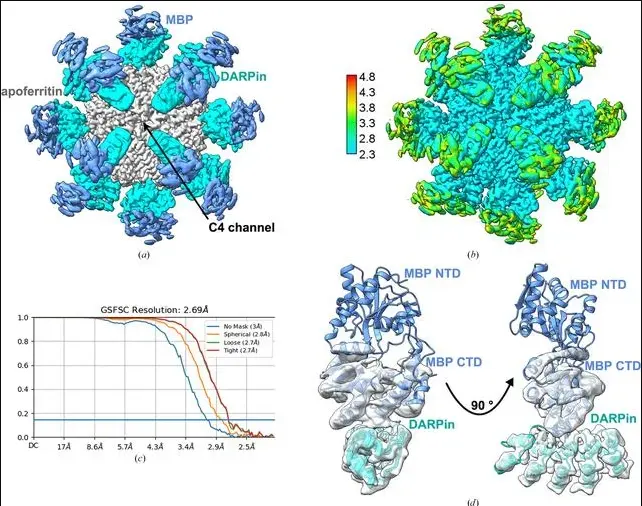
IUCr) A large, general and modular DARPin–apoferritin scaffold
enables the visualization of small proteins by cryo-EM
• Disulfide-Rigid Fabconstr, (Nature Communications, 2025): Using iterative molecular engineering, this design allowed for 2.3–2.5 Å resolution and provided a high-resolution cryo-EM structure.
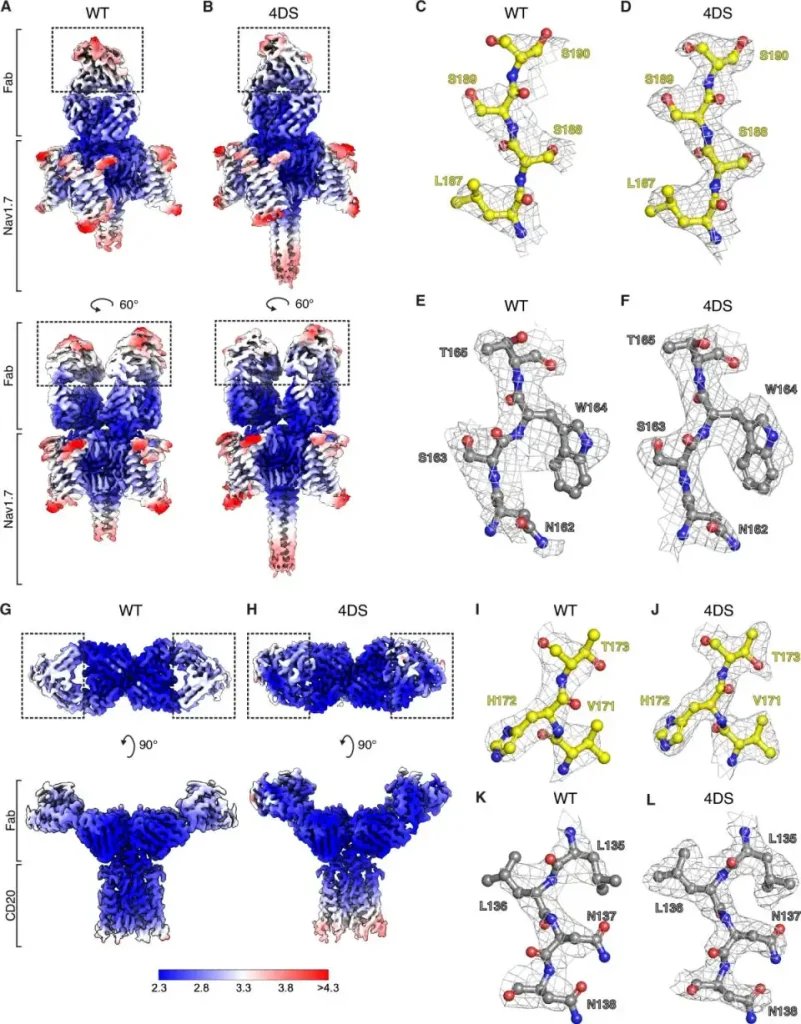
Disulfide-constrained Fabs overcome target size limitation for
high-resolution single particle cryoEM | Nature Communications
Advanced Instrumentation and Data Processing
Not all labs need scaffolding. For most well-behaved, low molecular weight membrane proteins (less than 100 kDa), determination of molecular structures is routine, thanks to advancements in instrumentation and data processing methods.
• Higher magnification targeting, thin ice. Increased magnification targeting thin ice sections can increase your sampling and lower data noise, respectively.
• Alignment improvement using 2D template matching. Alignment of low kDa (under 50 kDa) complexes is improved using 2D template matching with a well-resolved structure as a prior. The minimum kDa limit for single-particle cryo-EM is estimated to be around 38 kDa.
• Volta phase plate for contrast improvement. Phase plates that augment phase contrast of low spatial frequencies can facilitate the observation of smaller than the diffraction limit sized particles. The (52 kDa) surface streptavidin tetramer was resolved to 3.2 Å (Volta) phase plates, and thus exemplifies the value of phase plates for small sized specimen.
How Longlight Technology Supports Sub-100 kDa Cryo-EM Projects
At Longlight Technology, we understand that cryo-EM is a tool, not an end in itself—especially for small-protein targets where sample is limited and the path to structure is rarely linear. Our services are built around three principles that align with the needs of researchers tackling challenging low-mass targets:
• Transparent, Stepwise Workflows: Every project begins with sample suitability assessment via negative-stain examination to verify homogeneity, aggregation state, and particle morphology before committing to high-resolution data collection. This saves both time and valuable resources.
• Access to High-End Instrumentation: At our facility, we support cryo-EM and cryo-ET applications using the Glacios 2 (a 200 kV workhorse cryo-EM system optimized for routine single particle analysis) and the Titan Krios G4 (a 300 kV flagship platform designed to unleash the potential for both maximum stability and resolution). For the initial screening and evaluation, we also provide the Talos L120C G2, and offer teams the opportunity to assess sample behavior without excessive resource commitment.
• Complete Data Transparency: We provide all the raw cryo-EM movies, all the files from processed and unprocessed files, the final 3D density maps and the corresponding resolution and all atomic coordinates models (if present), and all cross validation reports. Full data availability ensures that your interpretation is never limited by what a service provider chooses to share.
Founded in 2015, Longlight Technology has focused on molecular diagnostics and structural biology, offering not only cryo-EM services but also precision laboratory instrumentation and genomic consumables such as focused ultrasonication systems and nucleic acid extraction kits. Our manufacturing expertise enables us to support researchers from sample preparation through final structural delivery—an integrated approach that is particularly valuable for small-protein projects where sample handling precision is critical.
Kết thúc
High-Resolution Cryo-EM for sub-100 kDa targets has moved from a frontier challenge to a solvable problem. Whether through mass-enhancement scaffolds, disulfide-constrained fragment antibodies, AI-designed rigid systems, or simply optimized data collection on modern instruments, the tools now exist to address the low-SNR barrier that has historically excluded small proteins from the cryo-EM revolution. As the global cryo-EM market expands and service providers like Longlight Technology make these tools more accessible, structural biology is finally catching up to the reality that small proteins are not peripheral—they are the majority.
Frequently Asked Questions
Q1: What is the minimum size limit for High-Resolution Cryo-EM today?
With optimized scaffolds (e.g., Di-Gembodies, Trimbody), data collection is effective down to ~14-20 kDa. A modern 300 kV instrument can solve proteins up to 50-70 kDa and without scaffolds.
Q2: Are scaffolds necessary for all sub-100 kDa constructs?
No. High-quality soluble proteins > 50 kDa can be solved without scaffolds. Poor SNR or proteins < 50 kDa are when scaffolds are the most useful.
Q3: What is the amount of sample needed in sub-100 kDa Cryo-EM?
For negative stain: ~100 µL at ~1 g/L. For high-resolution single-particle analysis, the sample amount in the same range is needed at the beginning, but grid optimization can necessitate additional material. Sample consumption is featured in Longlight Technology's workflow.
Q4: What is the expected resolution in case of a 50 kDa target with no scaffold?
Instrument choice affects data collection. For e.g. a Titan Krios G4 or a Glacios 2, resolution can be from 3.0 Å to 4.5 Å. A scaffold free sub-50 kDa range is challenging, and thus mass enhancement is the preferred solution.
Q5: Can I have a scaffold designed by Longlight Technology?
We're focused on data collection and assessment and transparent data processing. Specifically for scaffold engineering e.g. a nanobody, or with a DARPin, we would either support a client offer or work with an outstanding partner.
References:
Yi, G., Mamalis, D., Ye, M. et al. Covalently constrained ‘Di-Gembodies’ enable parallel structure solutions by cryo-EM. Nat Chem Biol 22, 69–76 (2026).
Kung, J. E., Johnson, M. C., Tegunov, D. et al. Disulfide-constrained Fabs overcome target size limitation for high-resolution single particle cryoEM. Nat Commun 16 (2025).
Trimbody with rigid AI-designed scaffolds enables atomic-resolution cryo-EM structure determination of small proteins. Nat Commun (2026).
A large, general and modular DARPin-apoferritin scaffold enables visualization of small proteins by cryo-EM. IUCrJ (2025).