bài liên quan
Phân tách thể tích lớn bảo tồn các phân tử sinh học nhạy cảm
2026-04-17Large Volume Separations are essential for producing vaccines, gene therapies, and recombinant proteins. Yet the conditions that enable scale—high flow rates, shear forces, and pressure—often degrade sensitive biomolecules. Proteins denature. DNA fragments. Viral vectors lose potency.
Intensifying Downstream Processing With Magnetic Separation
Traditional separation techniques were created for small molecules. Biologics are larger and much more sensitive than small molecules. Biologics require the separation techniques to preserve the integrity and the sensitivity of the biomolecule for the product to be useful. This article shows one of the many advantages of using a magnetic separation process, to the biomolecule, by showing the sensitivity of magnetic separation during Large Volume Separation.
Traditional Separation Methods
When most engineers consider the value of biomolecules lost after the separation process, they talk about the efficiency of the biomolecule-binding factor and the elution recovery. However, most degradation of the product happens prior to the yields being calculated and during the separation phase.
Traditional methods of separation expose molecules to numerous harmful physical and chemical forms during the separation stage.
•Separation of membranes and columns: The streams that separate the biomolecules cause folding, rotation, and rearrangement of the flexible structural and polymeric molecules, causing substantial and sometimes irreversible degradation.
•Separation through heating or cooling: Most traditional methods of separation require cycles of heating and cooling biomolecules. This leads to the destruction of the biomolecules and the formation of aggregates.
•Separation, that is pressure driven: Mechanical and tangential flow of biomolecules that are bound to the surface of the filter, can cause a fracture that is shear or irreversible once the biomolecule is lost.
•Separation through the use of elution buffers: Change in chemical surroundings and elution buffers of a lower (or sometimes higher) pH can alter the conformation of the biomolecule.
Dr. Lydia Kisley and her Case Western Reserve University team showed that some commercial separation materials labeled as “fully porous” have central sections that are mostly inactive. This means manufacturers are paying for the entire capacity, but are only receiving a small portion of the potential performance. This, combined with prolonged processing times, can lead to biomolecules degrading.
Intensifying Downstream Processing With Magnetic Separation
When taken cumulatively, the decreased specific activity, increase in biomolecule aggregation, and reduced final yields result in costly reprocessing.
Why Conventional Processing Methods Can't Develop Further
Laboratory methods have many separation methods that can be used, many that can become impractical on an industrial scale.
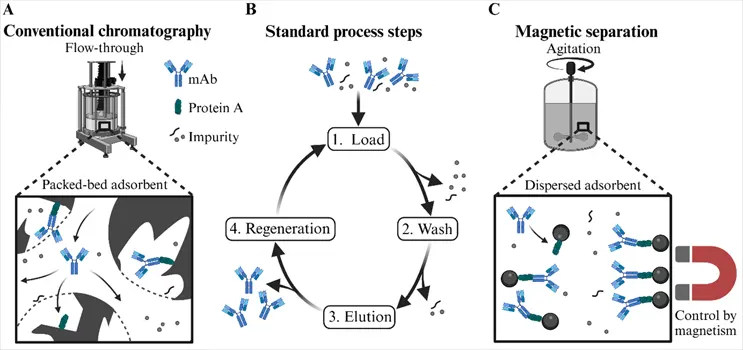
One example of a method with scaling problems is the use of chromatography columns.
•Narrowed transport of target: In packed-bed chromatography, binding occurs through the diffusion of an analyte. For biomolecules of sizes greater of 100 Da, the time of diffusion is near the binding surface and substantial area is excluded.
•Clogging: In order to prevent blockage in industrial-scale columns, the feed stream is prevented is pre-clarified.
•Increased buffer consumption: Industrial-scale columns create large-a mount of used buffer, which creates an increased cost to the operation.
•Shear sensitivity during packing and operation: The mechanical forces required to maintain uniform bed packing at scale can damage the very biomolecules the column is designed to purify.
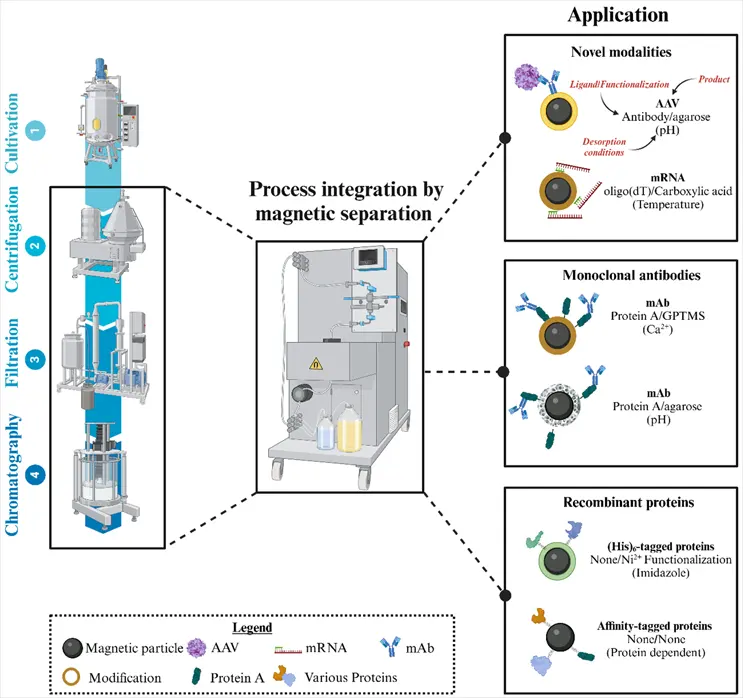
Researchers Julian Galbusera, Ines Zimmermann, and Paula Fraga-García of the Technical University of Munich have documented how magnetic separation technology addresses these bottlenecks. In contrast to standard chromatography, whereby the stationary phase is a matrix of packed beads permeated by a mobile fluid, magnetic separation processes the feed stream directly by suspending the functionalized magnetic particles. This allows the diffusion barriers that generally plague chromatography to be entirely circumvented, all while operating at large scales and processing unclarified lysates.
The Magnetic Alternative: Gentle, Fast, and Scalable
The principles of magnetic separation are fundamentally different. Functionalized magnetic particles target specific molecular species in suspension. The application of an external magnetic field captures the particle-target complexes and allows routine wash-out of undesired species. The particle-target species complex is shielded from shear and thermal stresses during processing.
For large volume separation processes, the advantages of this technique from the perspective of separation of sensitive biomolecules are several:
•No diffusion through pores is necessary: Magnetic particles are typically non-porous. Thus the slow diffusion step, which is a step that limits the binding kinetics of all chromatographic methods, is circumvented. It is worth mentioning that large biomolecules bind directly to the particle surfaces.
•Gentle capture and release: The magnetic field applies a soft volumetric force in contrast to the very high pressure and very high packing density that are characteristic of packed-bed columns. Cells remain intact. Viral vectors retain infectivity. Protein structures stay folded.
•Direct processing of crude loads: Magnetic separation can handle turbid, particulate-laden feed streams without pre-filtration. This eliminates one or more unit operations from the downstream train.
•Minimal buffer consumption: Because magnetic particles are suspended and retrieved rather than fixed in a column, buffer volumes can be dramatically reduced, lowering both cost and environmental impact.
A 2023 study published in Molecular Therapy—Methods & Clinical Development demonstrated the practical application of this principle. Researchers developed a filter-less magnetic capture method for purifying recombinant adeno-associated virus (rAAV5) directly from cell lysate. In under two hours, they achieved 63% recovery yield from approximately 5 liters of lysate, with a three-log reduction in host cell DNA and host cell proteins, while eliminating depth filtration and column chromatography steps entirely.
Real-World Performance mộtt Production Scale
The question for industrial manufacturers is not whether magnetic separation works in the laboratory—it is whether it performs reliably at production volumes. Evidence continues to mount in the affirmative.
•Liter-scale protein purification: Research published in ACS Omega demonstrated that a single-step magnetic fishing process could achieve 91% purity of green fluorescent protein (GFP) directly from crude Escherichia coli cell lysate at liter-scale volumes, using bare iron oxide nanoparticles produced via simple coprecipitation synthesis.
•High-gradient magnetic separation on a bigger scale: Many of the same high-gradient magnetic separation (HGMS) principles that allow lab purification are used in the production of industrial mRNA vaccines. One group created a 3D-printed, disposable HGMS separation chamber from a material compliant with USP Class VI. In this case, the chamber operated at flow rates of 150 mL per minute, with a retention exceeding 99.39%. This means that magnetic separation methods maintain capture efficiency at flow rates used in Large Volume Separations.
•Applications in cell therapy: Magnetic separation systems used to automate T-cell Isolation reach greater than 85% efficiency and greater than 96% purity, at cGMP ready scale for research and manufacturing, in 70 to 100 minutes.
Longlight Technology: Engineering for the Long Run
Separation technology that offers high-volume continuous service makes Longlight Technology's engineering focus.
In the realm of magnetic separation, Longlight's systems are designed to consistently maintain uniform magnetic field strength across large capture volumes - one of the most critical requirements for providing reproducible results batch after batch. Batch-to-batch reproducibility in Large Volume Separations is what regulated environments for manufacturing simply cannot tolerate.
The collection of systems provide answers to the specific challenges that large volume processing entails:
•Capturing magnetic forces highly concentrated across the entire capture zone: This refers to the problem of uneven distribution of a diminishing magnetic field gradient, whereby some particles are captured with high efficiency and others remain. They become captured and then escape, diminishing yield and providing variability.

•Separation chambers (vessels) capable of efficient capture as the volume of the chamber increases from a little above bench scale to production scale batch.
•Process flexibility: The system does not need to perform any significant modifications to accommodate differing types of magnetic particles, binders, and operating procedures.
Beyond Compromise
The balancing act of biomakers in choosing either efficiency in separation, or preservation of the biomolecule, is coming to an end. Technology that utilizes magnets in operations is not something that the industry is still testing. It is an established industrial practice and is substantiated in various peer-reviewed articles. These products can be found in the inventories of numerous undoubted producers and suppliers in the industry.
In applications where the value of the end-particle (gene closure, m RNA closure, monoclonal antibody closure, cell and cell products, etc.) is the distinguishing characteristic, magnetic separation:
1. Maintains biological activity that is lost in conventional separation;
2. Eliminates several separation operations by clarifying the initial feed.
3. Requires less buffer and less contact with the chemicals.
4.Iis easily modified from R&D to the full production line.
The scientific foundation is well-established. The engineering is mature. The question now is not whether magnetic separation belongs in large-volume bioprocessing, but how quickly manufacturers will adopt it to protect what matters most—the molecules that deliver therapeutic value.
To explore how magnetic separation systems can address Large Volume Separations in your specific process, visit www.longlight.com for technical specifications and application support.
FAQ
Q: Can a large volume of unclarified cell lysates be processed by magnetic separation?
A: Yes. Magnetic separation can be performed on feed streams with a high turbidity and does not require any feed stream pre-filtration, thus, reduces the number of unit operations.
Q: Is it true that protein activity suffers less degradation in biomagnetic separation?
A: Yes. Biomagnetic separation methods are not subject to high pressure/velocity or the use of severe buffer systems to separate and recover biomolecules.
Q: Can magnetic separation be scaled up from laboratory to commercial/industrial use?
A: Yes. The physics of magnetic capture operates in the same way in milliliters and hundred-liter systems of any volume.
Q: What biomolecules can be readily processed with magnetic separation in large volumes?
A: Viral vectors, mRNA, rProteins, exosomes, and cells, etc. These materials are susceptible to mechanical or thermal stresses.